US invests $232 million in at-domestic, phone-primarily based totally COVID-19 exams
Ellume will open a manufacturing unit inside the US to ramp up manufacturing.
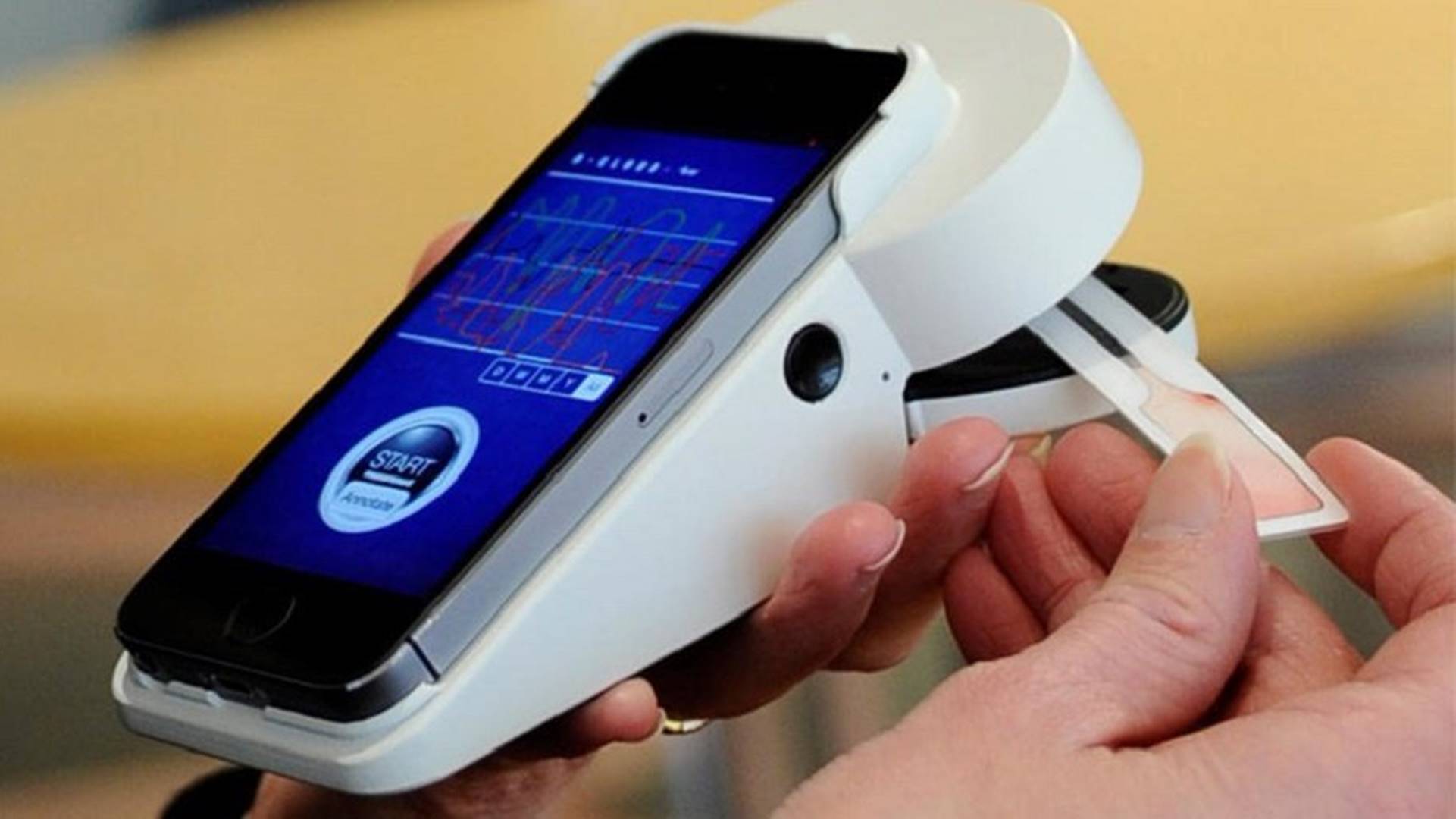
The US authorities are investing $231.eight million to boom manufacturing of an at-domestic COVID-19 check. Australian producer Ellume will supply eight. five million exams for federal use as a part of the deal. The agency will open a manufacturing unit inside the US, wherein it plans to make at the least 500,000 check kits in line with day via way of means of the quit of the year. Ellume is presently generating around 100,000 exams daily.
The Food and Drug Administration granted emergency clearance to Ellume’s check in December. The $30, a single-use check can examine a nasal swab and ship the consequences to an app in around 15 minutes. It’s stated to be ninety-five percentage accurate. As the Associated Press notes, Ellume’s check is one in all 3 that US citizens can self-administer and the handiest one it is to be had over the counter.
The husband and wife team behind the leading vaccine to solve Covid-19
Health specialists were calling for multiplied at-domestic checking out to make it less difficult for humans to decide whether or not they’ve COVID-19 and if so, to keep away from touch with others. However, it may take time for production to scale up.
Most exams nonetheless require fitness specialists to carry out a nasal swab, a lot of which can be despatched to labs for analysis. The FDA previously approved different exams that may supply consequences in below 15 minutes.
Many airways and countries require proof of a terrible COVID-19 check earlier than permitting vacationers to fly or input the country. Having the choice to perform a check at domestic with fast consequences may want to make it less difficult for humans to tour in the event that they want to.
Engadget / TechConflict.Com